“Life is looking up” – how a DBS stimulator upgrade changed Matt’s life
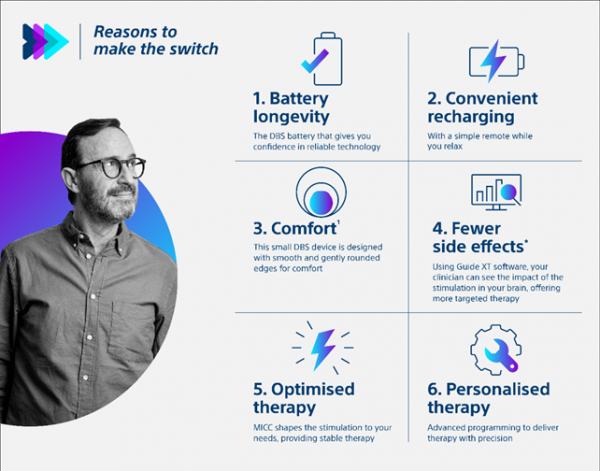
For patients with a Deep Brain Stimulation (DBS) device, switching to a new DBS battery can feel daunting. Yet, batteries don’t last forever and patients might experience a reduction in the effectiveness of the treatment over time. The good news: a replacement is not complicated and is done as an out-patient procedure.
For Matt Eagles, a Parkinson’s Disease patient who underwent the upgrade procedure for his DBS device, it is clear: "The switch has given me stability I've been craving for years." Matt received his original DBS device in 2006 and in 2020, he swapped his DBS device to one from Boston Scientific.
In such an upgrade procedure, the electrodes remain in the brain, and only the simulator placed in the abdomen is being exchanged – a shorter and simpler procedure compared to the first implantation. The Vercise M8 adapter enables linking existing electrodes to a Boston Scientific system, so patients can benefit from the latest technology. The new simulator gets then programmed to achieve optimal clinical results.
Since getting the upgrade, Matt has been able to achieve things that previously felt out of reach: “DBS has helped me tremendously. It energises me and it’s given me so much more confidence… I’m in control now. That’s a beautiful thing to have. I would say to someone that was hesitant about doing the switch… don’t be – it’s probably the best thing I've ever done!” As a self-declared adrenalin junkie, he invited us to join him for a ride on the fastest zipline in the world. Discover his story:
More on the upgrades here: https://www.bostonscientific.com/en-EU/upgrade-your-dbs-therapy.html
*Vitek JL, Jain R, Chen L, et al. Subthalamic nucleus deep brain stimulation with a multiple independent constant current-controlled device in Parkinson’s disease (INTREPID): a multicenter, double-blind, randomised, sham-controlled study. Lancet Neurology. 2020;19(6):491‐501. doi:10.1016/S1474-4422(20)30108-3.
The patient quotes in this material describe real personal experiences. Individual results may vary. Consult with your physician to determine if you are a candidate for this procedure and what you may gain from the therapy.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France. 2022 Copyright © Boston Scientific Corporation or its affiliates. All rights reserved.
NM-1346603-AA